Development and Scale Up Considerations for Nanosuspension Dosage Forms
Pharmaceutical companies continue to develop nano-formulations for poorly water-soluble drugs because of their proven effectiveness in commercial products. When considering and selecting a contract development and manufacturing organization (CDMO) to partner in this critical development pipeline, drug development teams must consider the CDMO’s experience, scientific team, and technologies.
There are two key reasons poorly water-soluble drugs are filling pharmaceutical pipelines:
♦ Enhance drug dissolution and oral bioavailability
♦ Increase drug loading and release duration for parenteral drug delivery. Preparation of drug nanosuspensions can be top-down, bottom-up, or a combination of both.
Drug development teams should initially concentrate on screening to identify the lead formulation that generates stable nanosuspensions that achieve the desired drug efficacy and pharmacokinetic properties in animals and humans. This is done during the nanosuspension formulation development process.
Once that is done, the nano-milling process should be optimized to determine the critical process parameters during scale up. Subsequently, CDMOs need to consider incorporating the nanosuspensions into the final market image dosage form based on the desired pharmaceutical product profiles of patients and available technologies.
Effective Planning Tools
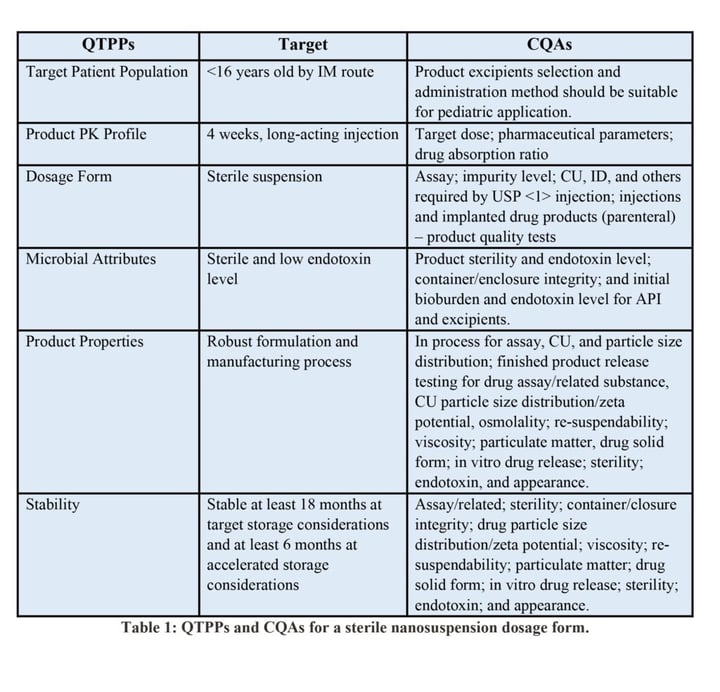
A Target Product Profile (TPP) is a planning tool for therapeutic candidates based on Federal Drug Administration (FDA) guidelines. Using the relationship between product quality attributes and product safety and efficacy profiles, a Quality Target Product Profile of Nanosuspensions (QTPPs) should be determined by the CDMO. It defines the desired profiles of the finished product and Critical Quality Attributes (CQAs). Table 1 lists QTPPs and CQAs for a sterile nanosuspension dosage form.
Nanosuspension Formulation Design
To maintain budget and timelines associated with developing nanosuspension formulations, CDMOs must carefully evaluate compound physical chemical and biopharmaceutical properties. Examples of those properties are solubility, pKa, solid surface properties, permeability, meting point, and crystal lattice structure.
A good candidate for nanosuspension has the characteristics of BCS Class II compounds, such as low solubility, high melting point, high permeability, and a strong tendency of food effects. Typically, the CDMO will conduct a small-scale formulation screening study to determine the suitable stabilizer(s). Among those evaluated are a polymer, surfactant, or polymer/surfactant combination for the nanosuspension.
Different surface properties of active pharmaceutical ingredient (API) may demand different types and levels of stabilizers. Nanosuspension performance, particularly stability, re-dispersibility and in vivo performance in animal models, should be confirmed by in vitro studies.
Stabilizer selection should be the first step in the development of the nanosuspension product. It can help prevent drug particles aggregation during milling and storage, and to steady the surfaces developed during the milling process. Stabilizers use a mixture of a polymer and an ionic/nonionic surfactant to stabilize drug suspension.
CDMOs must take careful consideration, especially related to stabilizer(s), milling speed and time, temperature, drug loading, bead size, and ratio of drug suspension to the milling medium. They all serve critical roles in the particle size distribution of the milled suspension.
Importance of QBD Design
A nano-milling process and scale up equipment utilized by Ascendia is shown in figure 1. The transfer of laboratory results for nano-milling from labs to production scale requires the scientific team to understand the critical process parameters. Specific energy input, stress energy of the grinding media, stress coefficient, and residence time distribution are among the factors.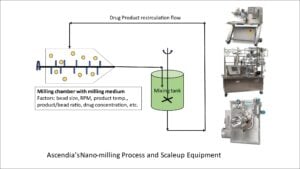
Formulation variables and process parameters, which may have a significant impact on the nanosuspension production, are all considerations when scaling is performed. Drug loading, stabilizer level, milling speed, time, temperature, grinding media size and density, suspending vehicle viscosity, volume of formulation to grinding media ratio, and percentage fill of milling chamber all must be considered.
CDMOs will conduct preliminary formulation screening studies in a lab milling system. Studies denote the size of the grinding media, the peripheral speed of the agitator, and the density of the grinding media. Each has an impact on wet-milling in an agitator bead mill.
Conclusion
Stabilizer concentration, drug loading, milling speed, milling time, bead diameter/density, temperature, and the amount of beads versus drug product are important formulation and process variables for drug nanosuspensions. Critical process parameters (CPPs) that impact nanosuspensions’ critical quality attributes should be identified using a DOE design. The ranking of CPPs impact on CQAs may change due to an alteration in milling design during the scale-up process. It is important that a design space of the CPPs be defined during the scale-up process in order to achieve a robust formulation and manufacturing process.
Contact us to learn how we can be your CDMO for complex injectables for early phase studies.
This Formulation Forum was originally published on Drug Development & Delivery.
