Nano-Emulsion Technology Creates New Methods to Overcome Poorly Soluble Drug Formulations
Drug development teams use emulsions to deliver active ingredients by multiple routes of administration, such as topical, oral, nasal, ophthalmic, or injecles. Scientists at pharmaceutical companies must decide which emulsion type – oil-in-water (O/W) or water-in-oil (W/O) – is best for their drug formulation. Making the correct choice can improve the chances for success, as well as time-to-market.
Oil-in-water emulsions have gained significant application in the delivery of lipophilic drugs or nutrition supplements by parenteral, oral, and topical routes of administration. Water-in-oil emulsions have increased their application for delivery of hydrophilic small molecules and biologicals, such as large molecules, DNA, RNA, and peptides.
Nano-size emulsions can provide unique solutions for overcoming drug solubility, permeability, and stability problems. They create these benefits because they can control droplet sizes much less than 100 nm, as well as a large surface area and a reduction in surface tension. There are two types of nano-sized emulsions: nano-emulsions and micro-emulsions. The distinction between the two relates to their thermodynamic stability. Each has its advantages and applications in specific drug formulations. A comparison of the emulsions is shown in Figure 1.
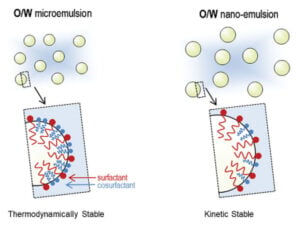
Figure 1
Characteristics of Micro-emulsions
A thermodynamic system, micro-emulsions require a high level of surfactant (greater than 50%). Micro-emulsions are thermodynamically stable due to the use of sufficient co-solvents and surfactants to prevent Ostwald ripening, which is essentially the coalescence of the droplets into larger particles. Ostwald ripening is the most frequent instability mechanism, although gravitational separation can also occur with larger particles. Many solvents and surfactants have undesirable side-effects. In fact, the Food and Drug Administration (FDA) places daily intake limits on such ingredients. For this reason, micro-emulsions are less desirable formulation options compared to nano-emulsions.
Nano-emulsion Considerations
Nano-emulsions form a kinetically stable system that only requires a low-level surfactant below 10%. Because nano-emulsions contain much less surfactants, they are meta-stable and more susceptible to Ostwald ripening. Nano-emulsions require greater kinetic formation energy, and are usually prepared using high-pressure homogenization, microfluidics, or ultrasonic generators. A well-formulated nano-emulsion, however, will maintain its physical-chemical stability through its typical 2-5 year shelf-life.
In addition, nano-emulsions possess benefits improving drug solubility; loading and stability; and reduction of injection pain and thrombophlebitiss. This enables targeted drug delivery and a marked reduction of drug toxicity. For those reasons, a number of lipid-based nano-emulsion pharmaceutical products have been marketed in the past few years, such as those in Table 1.

Table 1
Bioavailability Dosage Form Challenges
A drug’s low solubility often presents a serious challenge to developing bioavailable dosage forms. This hurdle can be exacerbated for drugs with chemical stability issues when solubility-enhancing approaches utilize excipients that are incompatible with the drug substance.
To overcome these obstacles, multiple methods have been developed:
Smaller particle size – Reducing the drug crystals to nanometer-size creates a greater surface area for dissolution
Production of amorphous solid dispersions – The energy required for dissolution is reduced due to amorphous solid dispersion
Lipid-based drug delivery systems – The ability to dissolve a hydrophilic drug in either a lipid or oil phase is achieved with these systems
Not all these technologies are suitable when the drug is poorly soluble and chemically unstable. In particular, the use of nano-emulsions is growing because of their characteristics and ability to develop poorly soluble drugs for multiple routes of administration. Examples include drops or creams for topical products, suspensions for pediatric and geriatric products, and sterile parenteral forms for injection. They can also potentially shield the active ingredients from chemical degradation, resulting in poor or sub-par product stability.
Even with their clear advantages, it must be noted that nano-emulsions do have limitations. The oil droplet particle size may increase over time via Ostwald ripening. The resulting physical instability can lead to a loss of optical clarity, which is deleterious and confounding for many ophthalmic products. Decreased drug solubility is also possible, as the interfacial surface area decreases.
To obtain physically stable nano-emulsions, long-chain triglyceride oils are sometimes employed. This approach usually requires organic co-solvents or toxic co-surfactants, which brings drawbacks, most notably safety concerns and a reduced tolerability profile of the pharmaceutical formulation. For these reasons, they are becoming less acceptable for use in pharmaceutical formulations.
EmulSol™ for Stable Nano-emulsions
With deep understanding of lipid chemistry and its interaction with drugs, and utilizing a tailored formulation approach, the Ascendia team developed a proprietary technology, EmulSol, for production of novel oil-in-water nano-emulsions.
Ascendia’s EmulSol technology produces stable, optically clear nano-emulsions without the use of organic solvents and with minimal surfactants using a high-pressure or microfluidic homogenization process. By selecting a specific lipid carrier with a surfactant and/or co-surfactant and a unique process, Ascendia has eliminated the use of organic solvents in its formulation approach.
EmulSol formulations are prepared using a robust, commercial-scale homogenization process that incorporates a proprietary combination of lipid carrier and surfactants. The resulting emulsion of oil droplets in the water phase is physically stable and safer for IV administration. Eliminating solvents from the formulation reduces injection site irritation and is more acceptable for pediatric, IV, and ocular products. Minimizing surfactants improves the safety and chemical stability of the resulting nano-emulsion formulation.
Ascendia has used its EmulSol technology to formulate its lead pipeline products. These include the ASD-002, a novel injectable form of the anti-thrombotic drug clopidogrel, and ASD-004, a clear emulsion cyclosporin eyedrop for dry eyes. Our next blog post will highlight how ASD-002 helped develop an injectable Clopidogrel for use in emergency situations.
Until then, feel free to contact us to find out how we can help in your drug development.
