Formulation Forum: Case Study in Lipid-based Nanoparticle Carrier Applications
A recent Formulation Forum column penned by Ascendia Founder and CEO Jim Huang, PhD, discussed how lipid-based colloidal nanocarriers administered by a parenteral route hold great promise in safely delivering therapeutic agents. The article was published on Drug Development & Delivery, a leading trade media property.
The guest editorial explains that the promise is due to lipid-based colloidal nanocarriers’ advantageous properties with respect to having excellent biocompatibility, potential for sustained drug delivery, ability to encapsulate hydrophilic and hydrophobic drugs, and site-specific active or passive targeting. Examples of lipid-based dosage forms include liposomes, lipoplexes, solid lipid nanoparticles, and nano-structure lipid carriers.
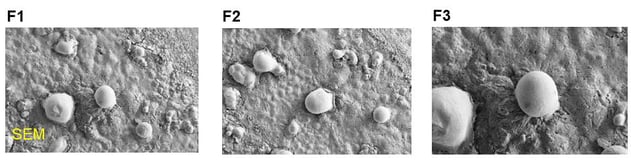
The opinion was supported by a study of parenteral sustained delivery of ASD-005 liposomal formulation. The study showed encapsulation of ASD-005 into a liposomal formulation with a nano-size range of 100 nm to 150 nm and with good physical stability. A differential scanning calorimeter (DSC) thermogram indicated that ASD-005 is present inside the liposomal formulation as an amorphous form.
The study indicated that parenteral administration of liposomal ASD-005 has a rapid onset effect of lowering hypertension followed by sustained release of the drug inside an animal body for at least 24 hours. Thus, the ASD-005 liposomal formulation avoids the rapid clearance that was experienced with an IV solution of the ASD-005 compound. Results suggest that the liposomal form of ASD-005 present an attractive sustained drug delivery for effective parenteral treatment of acute hypertension and congestive heart failure.
 You can read the entire column on the Drug Development & Delivery website. You can also contact us to learn more and discuss how Ascendia can help with your drug development pipeline.
You can read the entire column on the Drug Development & Delivery website. You can also contact us to learn more and discuss how Ascendia can help with your drug development pipeline.
